
GoGreen invites you to the HERIe workshops in Spain!
April 15, 2026By Qing Wu (HES-SO)
What is ‘sapoline’?
Sapoline is our newly developed green cleaning material for historical metal artefacts, particularly effective against corrosion on brass and steel.
The name “sapoline” comes from its two main components:
- Saponin: A naturally compound extracted from plants, which behaves like soap, lowering surface tension and binding to certain substances like heavy metals.
- Aquoline: A type of deep eutectic solvent (a special liquid mixture) made from natural components (choline chloride and water), considered eco-friendly and biodegradable. These two ingredients form an eco-friendly alternative to harsher chemicals.
Deep eutectic solvents (DESs) are made by mixing two substances in right proportions. When combined, they melt at a much lower temperature than either substance alone, which means they can be liquid at room temperature. They’re easy to make and are gaining attention as eco-friendly, “green” alternatives to traditional chemical solvents.
Initiative for the design of ‘sapoline’
One of my main research interests is finding green and sustainable ways to clean tarnished metals. Instead of relying on harsh chemicals, I’ve been exploring natural, bio-based materials drawing from both modern science and old recipes.
About a year ago, I came across soap nut decoction, historically used in German-speaking regions to clean lightly tarnished silver. When I tested a 10% solution on sterling silver coupons (artificially tarnished with boiled egg white), it worked well. But surprisingly, the same treatment failed on pure silver.
Analytical testing revealed why: sterling silver tarnish is rich in copper compounds, while pure silver tarnish is mainly silver compounds. Soap nut decoction, and specifically its active ingredient saponin, works better on copper-rich tarnish. Using ethylenediamine-N,N’-disuccinic acid (EDDS), a green copper-binding agent, confirmed this pattern.
This also explained why soap nut decoction worked well on some historical silver artefacts. Many of these objects were not of pure silver but of alloys containing copper. Copper makes silver more durable but also leads to more copper-rich tarnish over time.
These findings inspired the development of sapoline, a new, eco-friendly cleaning solution for metal alloys.
Development of ‘sapoline’
Brass was chosen for my initial experiments with saponin solutions, since it is the most widely used copper alloy. The results were promising: immersing artificially aged brass coupons in the solution, effectively remove the tarnish.
But immersion is not always practical. Many historical objects are delicate, so they require local application rather than full soaking. I also wanted to minimize the amount of saponin used. This led to the idea of a loading matrix, a material that can hold the solution and slowly release it onto a curved metal surface. Two loading matrices were then tested:
- Japanese paper: dried too quickly and couldn’t hold enough solution.
- Non-woven microfibre Evolon®: held more liquid but still dried within 15 minutes, giving only partial cleaning.
In one experiment, I tested a DES made by mixing ascorbic acid and choline chloride, which literature suggested should clean copper corrosion well. What caught my attention wasn’t just the cleaning effect: this DES kept Evolon® wet for a long time, allowing the cleaning agent to stay in contact with the metal surface much longer than water-based solutions. However, this DES turned out to be too aggressive and was very viscous. Testing other choline chloride-based DESs showed that choline chloride itself was responsible for the long working time, suggesting it could be paired with a mild cleaning agent. That’s when I returned to my earlier experiments with saponin.
The first prototype of sapoline was made by mixing a water solution of saponin with a well-known DES prepared from choline chloride and urea. This mixture cleaned tarnished brass surfaces quickly, effectively, and without being too aggressive. However, the Evolon® loading matrix turned green, which was different from Evolon® loaded with a pure saponin solution, where no colour change was observed. Such colour change indicated the formation of copper–chloride compounds (which usually appear blue-green in water) and the presence of free chloride ions.
Free chlorides can cause ongoing corrosion in metals. To address this, a second version of sapoline was developed using “aquoline” – a mixture of choline chloride and water with low viscosity. At a molar ratio of 1:3.33 (choline chloride:water), reported in the literature as a well-structured and stable system, aquoline proved to be a promising alternative. Three cleaning solutions were tested:
- Sapoline (saponin directly dissolved in aquoline)
- Low-aquo sapoline (less aquoline compared to sapoline)
- EDDS water solution
The results showed that sapoline’s cleaning power strongly depended on application time, while low-aquo sapoline was more sensitive to the saponin concentration. When limited to a maximum of 15 minutes on Evolon®, EDDS showed little cleaning effect. The comparison is illustrated in the figure below. Tests on corroded steel showed similar trends although cleaning was slower than on brass.
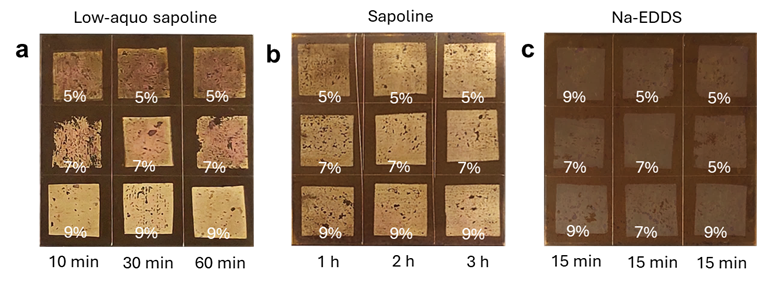
Figure 1: Cleaning of tarnished brass coupons with (a) Low-aquo sapoline, (b) Sapoline, and (c) EDDS water solutions. Reproduced from [Wu et al., 2025].
Further development
Research didn’t stop there. To enhance sapoline’s cleaning power, its preparation protocols were optimized to improve cleaning speed and ease of application. Sapoline-based gels were also developed, which clean effectively, absorb metal–saponin complexes, and offer high elasticity and strength compared to typical rigid gels.
Reference
Wu, Qing and Edith Joseph. “The application of an innovative green material ‘sapoline’ for the cleaning of historical metal objects”. Preprint for ICOMCC-Metal2025 Conference, 01-05 September, Cardiff

